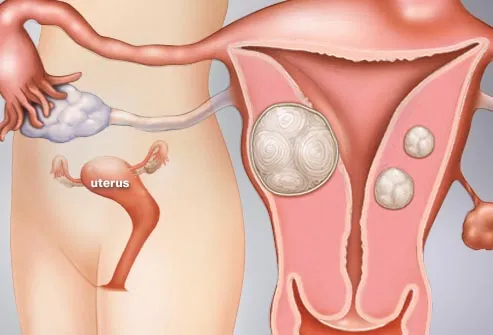
Leiomyomas of the uterus are one of the most common pathologic abnormalities of the female genital tract. Their occurrence increases with age, and they are found in 20–50% of women older than 30 years. Although found elsewhere in the body, leiomyomas most frequently occur in the myometrium. Uterine leiomyomas are commonly referred to as myomas, fibromyomas, or “fibroids” because of their firm, fibrous character and high content of collagen.
Leiomyomas of the uterus are the most common indication for hysterectomy in the United States. Approximately 175,000 hysterectomies are performed annually for leiomyomas. Uterine leiomyomas result in masses associated with a variety of gynecologic problems, the most prominent of which are the asymptomatic pelvic mass or abnormal vaginal bleeding.
The last two decades have seen rapid advancements in the diagnosis and treatment of uterine leiomyomas. The introduction of pelvic ultrasonography sonohysterography, hysteroscopy, and magnetic resonance imaging (MRI) have made it possible to detect small myomas not clinically suspected as a cause of abnormal bleeding. It is also possible to make a definitive diagnosis of a pelvic mass as a uterine myoma short of performing laparotomy or laparoscopy. Understanding the relation of sex steroid hormones and uterine leiomyomas has provided the foundation for using gonadatropin-releasing agonists (GnRH), which have facilitated treatment of leiomyomas by hysteroscopy, laparoscopy, and the liberal use of vaginal hysterectomy. With the introduction of the antiprogesterone RU486 and other drugs that can be used in lieu of surgery, the 21st century will be marked by dramatically new treatments of leiomyomas of the uterus.
SYMPTOMS
Uterine myomas are common, most are asymptomatic and require no treatment. Predominantly intramural or subserous, they are typically found in the highly screened U.S. population as unsuspected pelvic masses at the time of an annual examination and Pap smear.

Patients are usually between 30 and 50 years old. In the past, indications for intervention for asymptomatic uterine leiomyomas included uterine size, inaccessible ovaries, facilitation of estrogen replacement, and a rapid change in uterine size. A review of the literature does not support surgical intervention for uterine leiomyomas based on size alone. According to Mayer and Shipilov, normal ovaries that are not identified by ultrasonography can be differentiated from uterine leiomyomas on MRI, allowing exclusion of ovarian tumors.7 The addition of hormone replacement therapy in standard doses does not appear to provide sufficient amounts of estrogen or progesterone to cause uterine growth. Even the age-old accepted theory of a “rapidly enlarging uterus” has been challenged as an indication for intervention for the asymptomatic leiomyoma. Parker and coworkers reviewed the medical records of 1332 women admitted between 1988 and 1992 for surgical management of uterine leiomyomas. They found no correlation between rapid uterine growth and uterine sarcoma.8
Common symptoms associated with leiomyomas are menorrhagia, pelvic pressure or pain, and reproductive symptoms. The most common symptom associated with leiomyomas is menorrhagia. The cause of leiomyoma-associated menorrhagia is unknown. Ulceration of a submucous myoma and compression of venous plexus or adjacent myometrium and endometrium by intramural myoma have been postulated.9 Rarely, fibroids may present as a result of pressure on neighboring organs, such as urinary frequency or urinary obstruction secondary to urethral obstruction. Rectal symptoms also are rare, but blockage or irritation of the rectum and rectal sigmoid can occur. Acute pain associated with a low-grade fever and uterine tenderness may be observed with degeneration of leiomyomas or with torsion or a pedunculated subserous myoma.
The major clinical issues involved with leiomyomas of the uterus include the differential diagnosis, management of the asymptomatic pelvic mass, and appraisal of the role of the fibroid in various clinical problems such as abnormal bleeding, pain, infertility, and pregnancy. A review of current approaches to the differential diagnosis and treatment options for leiomyomas is appropriate.
TREATMENT
The problems associated with bleeding myomas are related primarily to anemia, but the myomas can also create difficulties in maintaining an acceptable lifestyle because of the problems of pelvic discomfort. The submucous myoma particularly can be the cause of major hemorrhage and produce emergency situations. Intramural fibroids and a distorted endometrial cavity can produce a similar picture and force intervention.
The clinical problems produced by the mass of the myomatous uterus usually are not observed until the mass rises out of the pelvis. In nonpregnant states, the mass may cause ureteral obstruction, bladder and rectal pressure, pelvic pain, and if the mass extends to the umbilicus or above, diaphragmatic pressure. Rarely, when the pedunculated subserous myomas twist and causes acute or chronic symptoms or marked abdominal protuberance, surgical intervention is warranted.
Benign degeneration of the myoma is usually caused by avascular necrosis. It is principally seen during pregnancy, when it should be treated symptomatically. Malignant degeneration results in sarcoma, which occurs in 0% to 5% of cases. Sarcomas arising de novo in normal uteri usually tend to be more aggressive. Changes in size, especially those associated with vaginal bleeding a postmenopausal woman, are signs of possible malignant degeneration.
Major advances in the treatment of symptomatic fibroids have created treatment options. The principal treatments for uterine myomas are control of the symptoms after a benign diagnosis is established. The options for treatment of symptomatic fibroids are medical management and surgery.
Medical Management
The management of symptomatic uterine myomas has traditionally involved surgery. However, the introduction of GnRH agonists in the 1980s offered temporary relief to women with myomas who do not desire surgery and are approaching menopause.18
The use of a GnRH agonist produces a hypoestrogenic state, which leads to a median reduction in uterine volume of approximately 50%. The maximal effect is seen after 12 weeks of therapy, with no further reduction in uterine size observed between 12 and 24 weeks of therapy. Unfortunately, rapid regrowth of myomas after therapy and side effects related to the hypoestrogenic state such as bone loss and hot flashes have limited the usefulness of the GnRH agonists.
To overcome the side effects of long-term GnRH therapy and maintain decrease in uterine size, Friedman and coworkers proposed an “add-back” regimen of estrogen and progesterone in small doses.19 Early reports with add-back therapy appear promising, but the high cost ($300.00/month) associated with GnRH agonist with estrogen and progesterone have limited this approach. The ultimate place for GnRH agonist therapy may be as a presurgical treatment before myomectomy or hysterectomy.
The GnRH agonist can cause a cessation of menses, allowing severe anemia to be corrected before surgery and avoiding preoperative blood transfusions. It also been shown to reduce operative blood lose. Preoperative GnRH also opens the possibility for converting some cases of abdominal hysterectomy into a vaginal hysterectomy through reduction in uterine mass.
Surgical Management
The key factors governing the surgical options are the patient's age, fertility status, and reliability; the impact of fibroids on surrounding organs; and attendant medical, surgical, or anesthetic risks that may complicate surgical treatment. If fertility is a consideration and the patient has major symptoms, myomectomy is the procedure of choice. Myomectomy should be preceded by a hysterosalpingogram to determine the location and patency of the fallopian tubes and to screen for submucous myomas. Extensive and aggressive myomectomy runs the risk of resulting in emergency hysterectomy. The use of uterine tourniquets and vasopressin has reduced the amount of blood loss during myomectomy. Dillon reported that, with the use of vasopressin, 72% of patients undergoing myomectomy did not need blood products.20 Other complications associated with abdominal myomectomy are pelvic adhesion and bowel obstruction. In comparing the overall morbidity associated with myomectomy with that of hysterectomy, Iverson found no difference.21
A patient undergoing myomectomy should understand that the procedure is a treatment for fibroids, not a cure. There is a 15% recurrence rate of myomas after myomectomy and a 10% reoperation rate.
Dubuisson first reported myomectomy under laparoscopic control.22 Technical innovations such as electric morsulators (Karl Stortz) and simplified suturing techniques have made this procedure more feasible. Laparoscopic myomectomy can be offered as an alternative to abdominal myomectomy in selected cases (i.e. with a single myoma smaller than 6 cm) and should only be attempted by experienced laparoscopic surgeons. It has been suggested that adequate closure of the myometrium after laparoscopic myomectomy is difficult and may account for uterine rupture during pregnancy after undergoing a laparoscopic myomectomy.